Bases may be used to remove contaminating humic acids. Acids may be used to eliminate contaminating carbonates. 
Pre-treatment seeks to remove from the sample any contaminating carbon that could yield an inaccurate date. The objective of pre-treatment is to ensure that the carbon being analyzed is native to the sample submitted for dating. 
Most samples require chemical pre-treatment to ensure their purity or to recover particular components of the material. Since there are practical limits to the age range of the method, most samples must be younger than 50,000 years and older than 100 years. AMS technology has allowed us to date very small samples (such as seeds) that were previously undatable. Modern AMS (accelerator mass spectroscopy) methods require tiny amounts, about 50 mg. What can be dated?Īny organic material that is available in sufficient quantity can be prepared for radiocarbon dating. However, to avoid confusion all radiocarbon laboratories continue to use the half-life calculated by Libby, sometimes rounding it to 5570 years. Subsequent work has shown that the half-life of radiocarbon is actually 5730 ± 40 years, a difference of 3% compared to the Libby half-life. After this time, there is little if any c14 left. The diminishing levels via decay means that the effective limit for using c14 to estimate time is about 50,000 years. 
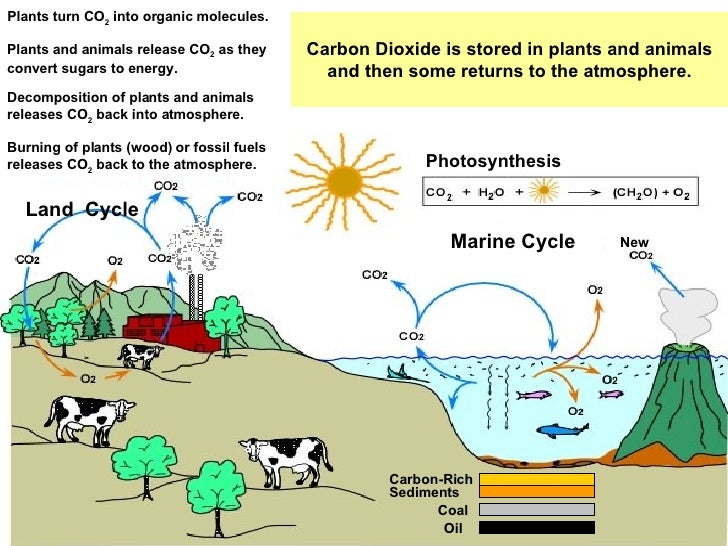
This means that half of the c14 has decayed by the time an organism has been dead for 5568 years, and half of the remainder has decayed by 11,136 years after death, etc. Libby calculated the half-life of c14 as 5568 ± 30 years. The measurement of the rate of radioactive decay is known as its half-life, the time it takes for half of a sample to decay. This is the clock that permits levels of c14 in organic archaeological, geological, and paleontological samples to be converted into an estimate of time. However, when the organism dies, the amount of c14 declines such that the longer the time since death the lower the levels of c14 in organic tissue. During the lifetime of an organism, the amount of c14 in the tissues remains at an equilibrium since the loss (through radioactive decay) is balanced by the gain (through uptake via photosynthesis or consumption of organically fixed carbon). Plants take up c14 along with other carbon isotopes during photosynthesis in the proportions that occur in the atmosphere animals acquire c14 by eating the plants (or other animals). The ensuing atomic interactions create a steady supply of c14 that rapidly diffuses throughout the atmosphere. What is radiocarbon dating?Ĭarbon-14 is produced in the upper atmosphere when cosmic rays bombard nitrogen atoms. Whereas carbon-12 and carbon-13 are stable isotopes, carbon-14 is unstable or radioactive.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
